Probably you’ve seen the Periodic Table of Heavy Metal over at IFLS. http://on.fb.me/M8CJrh
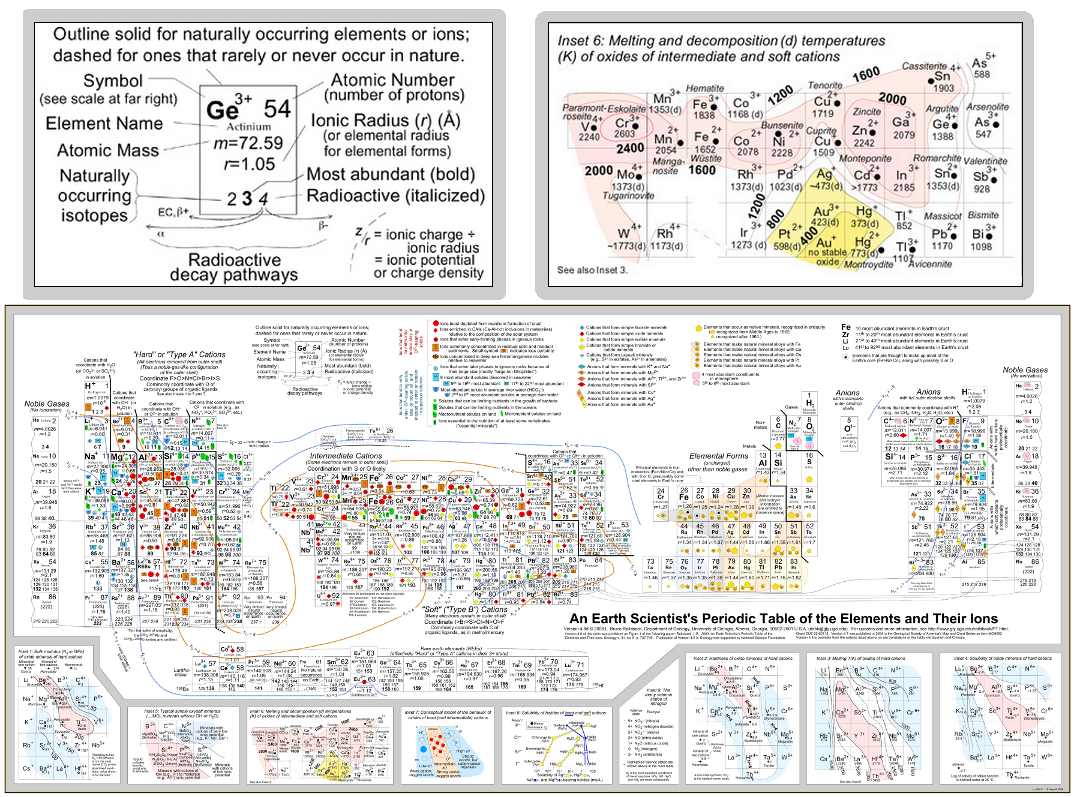
Although I’m quite a metal enthusiast, I have to say this doesn’t quite match the most awesome periodic table I’ve seen. It’s the Earth Scientist’s Periodic Table of Elements and Their Ions.
There is a popup-version of it with a lot of help to work you way through the periodic table. Our picture will just help you make the start to dig right in.
You can see it very well resembles the Period Table you know, but there is some more information that the geochemist among us find useful. Additionally to the Symbol, its atomic number it gives the element name, its mass, ionic radius in ?ngstr?m, as well as the naturally occurring isotopes with information about abundance and radioactivity. Plus, if you look at the radioactive isotopes it shows the decay pathways of that isotope.
If you want to get really into it, the element symbol also shows the relative abundance of elements in form of font size. You can see some colored lines across the entire Table, well these are contour lines marking the isopotential (This means elements of equal charge density.)
On the right side our little mash-up shows one of the many insets. This is inset 6 of 9 and shows the decomposition or melting temperature (in Kelvin) of intermediate and soft cations. Most will probably know that Mercury (Hg) is quite easy to melt (below 0?C at ?38,36?C), accordingly the decomposition contour line marks the lowest area of the inset.
Maybe you stumbled over hard, intermediate and soft cations. I did too and found the explanation that geochemists classify cations according to their bonding mechanism, be it ionic (hard) or covalent (soft). These soft cations are more prone to leaving their bond in a solution. If you want to have a closer look at this, follow the link.
Of course the periodic table features anions as well and also shows which cations and anions are likely to form minerals. Additionally, uncharged elements and noble gases are featured, but see for yourself!
See full Periodic Table: http://bit.ly/OziB1D
Slide on categorizing cations: http://bit.ly/OzpqAc
-JSD
via The Earth Story’s Facebook Wall
Jesper Dramsch
Latest posts by Jesper Dramsch (see all)
- Juneteenth 2020 - 2020-06-19
- All About Dashboards – Friday Faves - 2020-05-22
- Keeping Busy – Friday Faves - 2020-04-24